Columbia University
Irving Medical Center
Neurological Institute
710 West 168th Street, 3rd floor
(212) 305-1818
TaubCONNECT Research Perspectives:
March 2019
2: » FDG-PET Patterns Associated with Underlying Pathology in Corticobasal Syndrome
 Tae-Wan Kim, PhD
Tae-Wan Kim, PhDFamilial Alzheimer’s disease (FAD)-associated presenilin 1 (PS1) serves as a catalytic subunit of γ-secretase complex, which mediates the proteolytic liberation of β-amyloid (Aβ) from β-amyloid precursor protein (APP). In addition to its proteolytic role, PS1 is involved in non-proteolytic functions such as protein trafficking and ion channel regulation. Furthermore, postmortem AD brains, as well as AD patients, have shown dysregulation of cholesterol metabolism. Since cholesterol has been implicated in regulating Aβ production, Taub faculty member Dr. Tae-Wan Kim, in collaboration with Dr. Sungkwon Chung and team from Sungkyunkwan University School of Medicine in South Korea, investigated whether the FAD PS1-associated cholesterol elevation could influence APP processing.
As recently published in PLOS ONE, they found that in Chinese hamster ovary (CHO) cells stably expressing FAD-associated PS1 ΔE9, total cholesterol levels are elevated compared to cells expressing wild-type PS1. They also found that localization of APP in cholesterol-enriched lipid rafts is substantially increased in the mutant cells. Reducing the cholesterol levels by either methyl-β-cyclodextrin or an inhibitor of CYP51, an enzyme mediating the elevated cholesterol in PS1 ΔE9-expressing cells, significantly reduced lipid raft-associated APP (see Fig. 4). In contrast, exogenous cholesterol increased lipid raft-associated APP. These data suggest that in the FAD PS1 ΔE9 cells, the elevated cellular cholesterol level contributes to the altered APP processing by increasing APP localized in lipid rafts. This is the first report demonstrating that elevated cholesterol owing to presenilin FAD could have functional impact on APP processing by modulating lipid raft localization of APP.
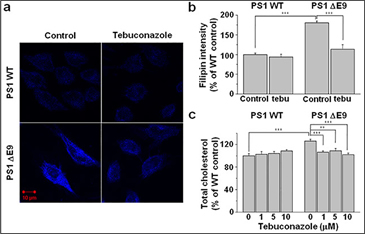 | Figure 4. Elevated cholesterol level in CHO PS1 ΔE9 cells was decreased by the inhibition of cholesterol biosynthesis.
(a) CHO PS1 WT and ΔE9 cells were pre-treated with 1 μM tebuconazole for 48 h, and free cholesterol was visualized by filipin staining. (b) Filipin intensities were compared in the absence (control) and presence of tebuconazole (tebu). PS1 WT control (n = 14), PS1 WT with 10 μM tebuconazole (n = 11), ΔE9 control (n = 10), ΔE9 with 10 μM tebuconazole (n = 10). (c) CHO PS1 WT and ΔE9 cells were pre-treated with 0, 1, 5, or 10 μM tebuconazole for 48 h, and total cholesterol level was measured from cell membrane fractions using Amplex Red cholesterol assay kit (n = 8). One way ANOVA: **p<0.01, ***p<0.001. |
Tae-Wan Kim, PhD
Associate Professor of Pathology and Cell Biology (in the Taub Institute)
twk16@cumc.columbia.edu
FDG-PET Patterns Associated with Underlying Pathology in Corticobasal Syndrome
 |  | |
| Edward D. Huey, MD | William C. Kreisl, MD |
The clinical diagnosis of corticobasal syndrome (CBS)—a disorder characterized by asymmetric parkinsonism, apraxia, cortical sensory deficits, and cognitive impairment—has been associated with different neuropathologic substrata, including corticobasal degeneration (CBS-CBD), Alzheimer disease (CBS-AD), and progressive supranuclear palsy (CBS-PSP). Clinical features are insufficient to identify the underlying pathologic substrate of CBS with certainty during life. 18Fluorodeoxy-glucose PET (FDG-PET) has been used to aid in CBS diagnosis, but the use of FDG-PET to try to differentiate the underlying pathologic substrata of CBS in vivo has not been extensively explored, even if available evidence suggests that different pathologies might show distinctive patterns of brain metabolism in CBS.
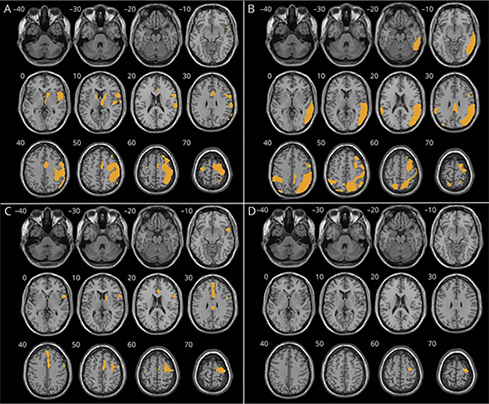
Figure 3: Regional hypometabolism compared to controls in the corticobasal syndrome (CBS)–corticobasal degeneration (CBD) (A), CBS–Alzheimer disease (B), and CBS–progressive supranuclear palsy (C) cohorts.(D) Conjunction analysis among the 3 groups. Statistical significance set at p < 0.05 family-wise error–corrected for multiple comparisons, except for C, where the threshold is p < 0.0001 uncorrected. Minimum cluster size: 100 voxels. The right side of the image represents the hemisphere contralateral to the more severely affected limbs. Significant clusters overlaid on a volumetric brain MRI template.
Thus, in collaboration with colleagues from multiple institutions, Taub faculty members Drs. Edward (Ted) Huey and William (Chuck) Kreisl co-authored a new study that systematically evaluated differences in topography of gray matter hypometabolism among different neuropathologic diagnoses underlying a CBS diagnosis, as assessed with FDG-PET and postmortem neuropathologic examination. As published in Neurology, they found that the primary pathology significantly affected the distribution of FDG-PET metabolism, with typical posterior temporoparietal or medial–frontal patterns observed in patients with CBS with a pathologic diagnosis of AD or PSP, respectively, and frontoparietal cortex, thalamus, and caudate nucleus in CBS-CBD. Their study demonstrates FDG-PET as an effective, complementary tool for proteinopathy identification in neurodegenerative diseases.
Edward D. Huey, MD
Associate Professor of Psychiatry and Neurology (in the Taub Institute)
edh2126@cumc.columbia.edu
William C. Kreisl, MD
Boris and Rose Katz Assistant Professor of Neurology (in the Taub Institute)
wck2107@cumc.columbia.edu